Menu
Molar mass of Fe = 55.845 g/mol
Convert grams Fe to moles or moles Fe to grams
| Symbol | # of Atoms | Iron | Fe | 55.845 | 1 | 100.000% |
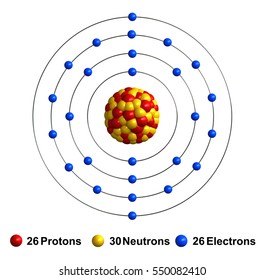
Here, we report a Fe single‐atom electrocatalyst supported on low‐cost, nitrogen‐free lignocellulose‐derived carbon. The extended X‐ray absorption fine structure spectra confirm that Fe atoms are anchored to the support via the Fe‐(O‐C 2) 4 coordination configuration. Of a single (Fe) atom along the edge of single-layer graphene is directly related to the atomic configuration of the graphene edge. Moreover, the random 1D diffusion of an Fe atom along a graphene. Besi adalah unsur kimia dengan simbol Fe (dari bahasa Latin: ferrum) dan nomor atom 26. Merupakan logam dalam deret transisi pertama. Ini adalah unsur paling umum di bumi berdasarkan massa, membentuk sebagian besar bagian inti luar dan dalam bumi. Iron is a metal in group VIII of the periodic table with atomic number 26, an atomic weight of 55.85, and a density of 7.86. It has a melting point of 1539 C. The electronic configuration of the Fe atom is (Ar) (3d) 6 (4s) 2, and it has an atomic radius of 0.126 nm. The radius of Fe 2+ is 0.076 nm, and that of Fe 3+ is 0.064 nm. Electronic States and Coordination Environment of Fe in Single-Fe-Atom Catalysts The 57Fe Mo¨ssbauer spectroscopy, which is highly sensitive for probing the oxidation state, electron spin configuration, and coordination environment,45–47 was carried out to determine the electronic states and coordination environment of Fe in single-Fe-atom catalysts.
In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.
Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.
Formula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. These relative weights computed from the chemical equation are sometimes called equation weights.
A common request on this site is to convert grams to moles. To complete this calculation, you have to know what substance you are trying to convert. The reason is that the molar mass of the substance affects the conversion. This site explains how to find molar mass.
The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. We use the most common isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.
Finding molar mass starts with units of grams per mole (g/mol). When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.
Teletype for Atom
Iron Atomic Model
Great things happen when developers work together—from teaching and sharing knowledge to building better software. Teletype for Atom makes collaborating on code just as easy as it is to code alone, right from your editor.
Share your workspace and edit code together in real time. To start collaborating, open Teletype in Atom and install the package.
GitHub for Atom
A text editor is at the core of a developer’s toolbox, but it doesn't usually work alone. Work with Git and GitHub directly from Atom with the GitHub package.
Create new branches, stage and commit, push and pull, resolve merge conflicts, view pull requests and more—all from within your editor. The GitHub package is already bundled with Atom, so you're ready to go!
Everything you would expect
Cross-platform editing
Atom works across operating systems. Use it on OS X, Windows, or Linux.
Built-in package manager
Search for and install new packages or create your own right from Atom.
Smart autocompletion
Atom helps you write code faster with a smart and flexible autocomplete.
File system browser
Free download opera gx browser. Easily browse and open a single file, a whole project, or multiple projects in one window.
Iron Atom
Multiple panes
Stylecop visual studio 2019 download. Split your Atom interface into multiple panes to compare and edit code across files.
Find and replace
Find, preview, and replace text as you type in a file or across all your projects. Munchymc.
Make it your editor
Packages
Choose from thousands of open source packages that add new features and functionality to Atom, or build a package from scratch and publish it for everyone else to use.
Fe Atom Electron Configuration
Themes
Atom comes pre-installed with four UI and eight syntax themes in both dark and light colors. Can't find what you're looking for? Install themes created by the Atom community or create your own.
Customization
It's easy to customize and style Atom. Tweak the look and feel of your UI with CSS/Less, and add major features with HTML and JavaScript.
See how to set up Atom
Under the hood
Atom is a desktop application built with HTML, JavaScript, CSS, and Node.js integration. It runs on Electron, a framework for building cross platform apps using web technologies.
Open source
Atom is open source. Be part of the Atom community or help improve your favorite text editor.
Keep in touch
| GitHub | github.com/atom |
| @AtomEditor | |
| Chat | Slack |
| Forum | Discuss |
| Stuff | Atom Gear |
| RSS Feed | Packages & Themes |
